USP <1062> TABLET COMPRESSION CHARACTERIZATION
Gamlen Instruments is your USP <1062> testing partner!
WHY WE NEED COMPACTION DATA
“Powder compression is a critical process in manufacturing the tablet dosage form. Although this process has been used routinely for over a century, problems related to powder compression in pharmaceutical formulation development and manufacturing persist. Common problems include tablet failures, such as capping and lamination, high friability, powder sticking to punch surfaces or the die wall, and insufficient mechanical strength to withstand stress in downstream processing.”
USP <1062>Tablet Compression Characterization
The compaction triangle illustrates the relationships between tabletting parameters. USP <1062> recommends generating all compaction triangle data for compaction characterization. The D- and R-Series instruments are supplied with the Gamlen Dashboard software which automatically calculates, analyses and interprets all compaction triangle data. Our instruments will allow you to not only comply with the USP, but also help you use that data to;
- assess your formulations prior to manufacturing
- troubleshoot manufacturing problems
- compare compaction behaviour of materials from different suppliers
- establish the intrinsic compaction behaviour of your APIs
- support change control activities
- reduce costs, limit material wastage and save time!

COMPACTION PRESSURE
The compaction force is the force applied to the powder bed. Compaction pressure is calculated from compaction force to account for the tablet dimensions. The applied area for a cylindrical tablet is the area of a circle calculated from the radius of a tablet face.
SOLID FRACTION
The tablet solid fraction quantifies how much of the tablet is solid i.e. not porous. It is calculated from the apparent or envelope density of a tablet divided by the material true density. It is the inverse of porosity (Solid fraction = 1 − porosity).
TENSILE STRENGTH
Tablet breaking strength is the force required to cause tablet fracture. Tensile strength or tensile fracture stress is calculated from breaking strength to account for tablet dimensions. It is calculated based on diametral compression testing as follows;
COMPRESSIBILITY
ARE YOU AT RISK OF OVER-COMPACTION?
The compressibility profile shows how the solid fraction of the tablets change with increasing compaction pressure. The solid fraction of a tablet is the inverse of its porosity. A tablet with a high solid fraction will have a low porosity e.g. a tablet with solid fraction of 0.9 has a porosity of 0.1. High solid fraction values are associated with capping problems and can result in slow tablet dissolution.

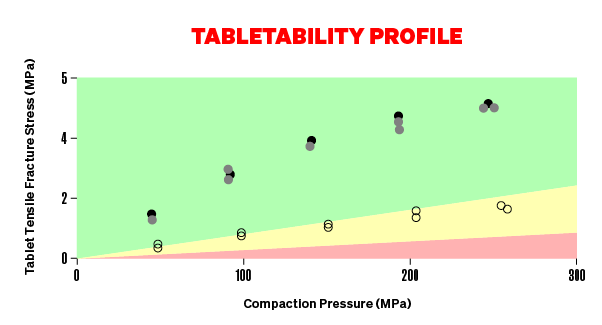
TABLETABILITY
ARE YOUR TABLETS TOO WEAK?
The relationship between tablet tensile strength and compression pressure is termed “tabletability”. To produce robust tablets that can withstand handling and transport they need to have sufficient tensile strength. It usually increases initially with increasing compaction pressure. It can then continue to increase, gradually level off or decrease at higher pressures. A decrease in tablet strength with increasing pressure is referred to as over-compaction and it is the result of tablet defects occurring at higher compression pressures. This often leads to serious manufacturing problems as the tendency is to increase compaction pressure when the desired tablet hardness is not being achieved. For this reason, it is essential that you characterise the tabletability of a formulation to avoid manufacturing problems. Using the Gamlen Dashboard, you can easily characterise the tabletability of your APIs, excipients and formulations.
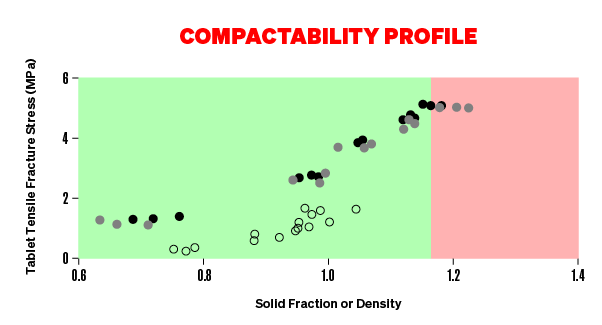
COMPACTABILITY
IS YOUR FORMULATION OPTIMISED?
The relationship between table tensile strength and solid fraction (or porosity) is termed compactibility. Typically, tensile strength will increase exponentially as solid fraction increases. Low solid fraction weakens a tablet but over-compaction to very high solid fractions introduces flaws into a tablet reducing its strength. By carrying out compaction analysis with a Gamlen instrument you will be able to make better tablets that are both strong enough to be robust but porous enough to be soluble.

UNDERSTANDING POWDER COMPACTION ANALYSIS
Interested in learning more?
Enjoy an insightful webinar hosted by Gamlen’s Product Director, Dr. Rebecca McVicker, where you can learn more about the science and method behind effective powder compaction analysis and see our compaction analysis suite in action.
