Compaction and tableting properties of composite particles of microcrystalline cellulose and crospovidone engineered for direct compression.
Here at Gamlen we are highlighting some of the fascinating and ground-breaking research that has been carried out by our customers in our Paper of the Month posts.
In this paper the authors investigate the compaction behaviour of a co-processed excipient (CPE) using a Gamlen Powder Compaction Analyser.
- Co-processing, a particle engineering technique, was used to produce composite particles of microcrystalline cellulose (MCC) and crospovidone (CPV).
- Compaction analysis was carried out on the MCC, CPV and the co-processed particles using a Gamlen.
- Heckel and Kawakita plots were generated for each material as well as tabletability, compactibility and compressibility data.
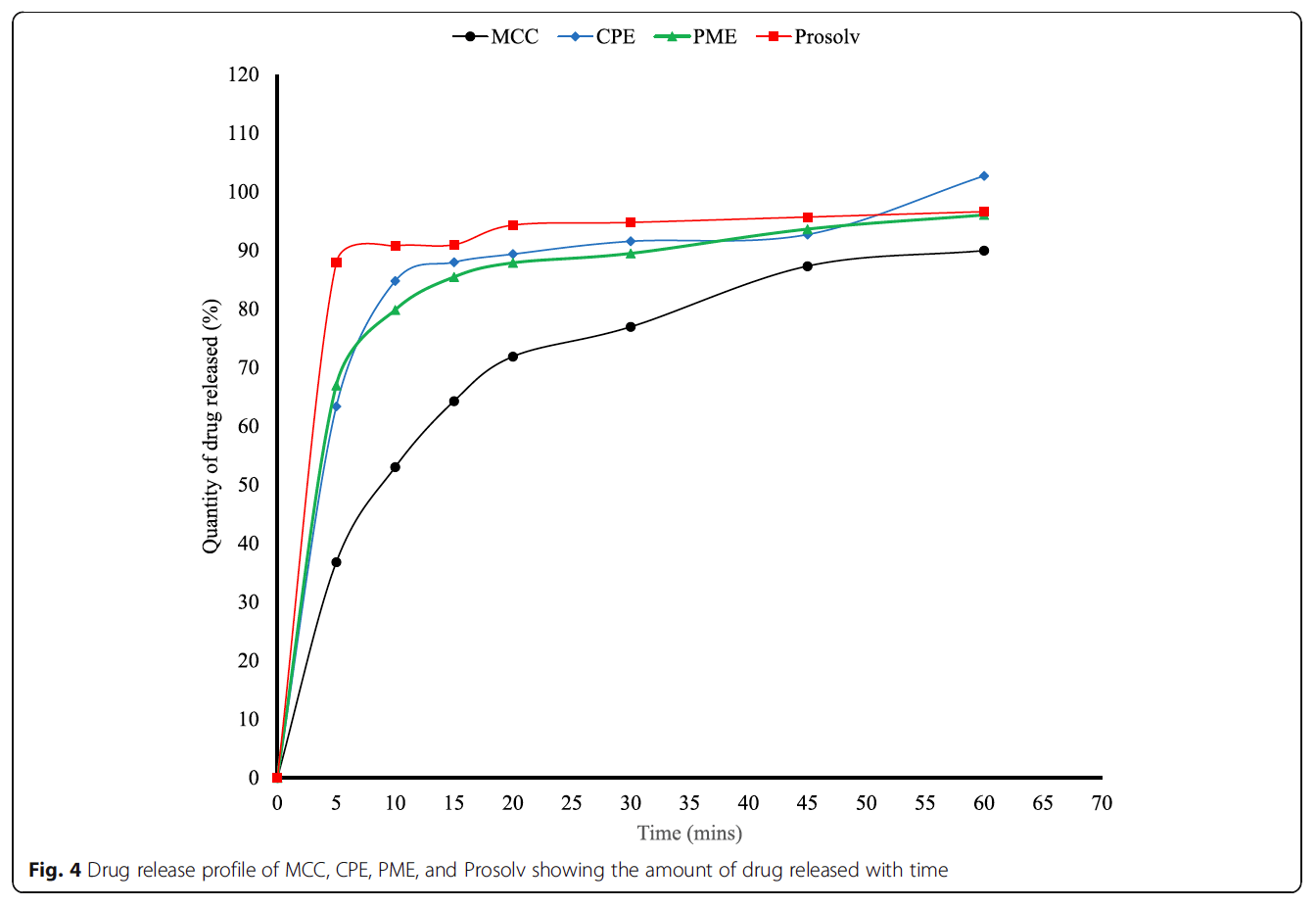
- Formulated tablets were produced by direct compression from MCC and the co-processed particles using metronidazole as a model drug.
- The lubricant sensitivity and the dilution potential of the co-processed excipient was assessed and the properties of the formulated tablets were evaluated.
CONCLUSION
The co-processed particles yielded tablets with enhanced dissolution and drug release profiles compared to MCC. This study illustrates the potential to use co-processing to improve the tableting properties of excipients.
Interesting in learning more about the work that is being carried out by the Gamlen community?
Join us on Thursday, June 10th, for our Virtual User Group.
Four speakers will discuss topics related to their experience of using of Gamlen instruments. It will be an opportunity to gain a deeper understanding of the benefits of compaction analysis and the applications of Gamlen Instruments. Register for our FREE Gamlen User Group online event today.







Leave A Comment