The evaluation of dynamic compaction analysis as a QBD tool for paediatric orodispersible minitablet formulation
Here at Gamlen we are highlighting some of the fascinating and ground-breaking research that has been carried out by our customers in our Paper of the Month posts.
In this paper the authors use a Gamlen D1000 to carry out compaction analysis and show how this compaction data can be used as part of a Quality by Design strategy in the formulation of orodispersible minitablets.
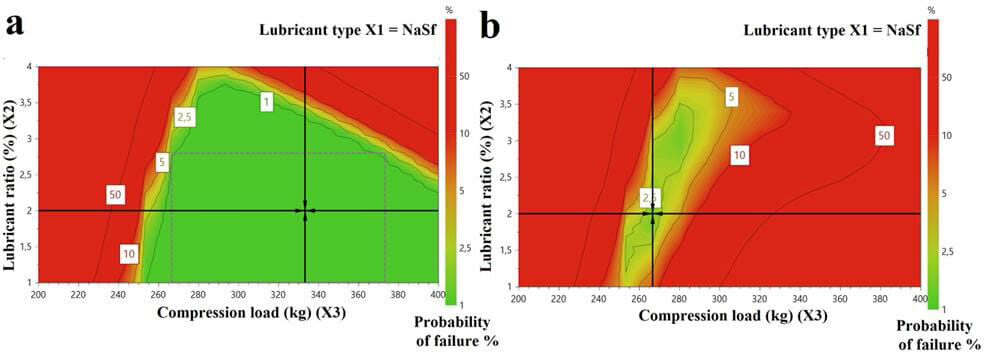
Design Space for the placebo orodispesible tablets that meet the specifications for disintegration time and crushing strength (a) and for compressibility index, detachment stress, ejection stress, disintegration time and crushing strength (b), expressed as probability of failure as a function of the lubricant ratio (X2) and compression load (X3)
- The authors began by identifying the key critical quality attributes (CQAs) of the product and how they related to formulation and process parameters.
- A design of experiments was developed for a better understanding of the effects of lubricants and of the compression force on the powders’ and tablets’ characteristics
- Data from the DoE was used to establish a Design Space (DS) to ensure both the quality of the tablets and the smooth running of the compression.
- ODMTs were prepared using two formulations, one within and one outside the design space. It was found that compression errors could be avoided by keeping the process within the limits of the design space.
CONCLUSION
These results suggest that the parameters derived from the dynamic compaction analysis could be useful from the early phases of the development to predict and avoid further manufacturing errors and material loss. The authors found that their strategy “fits perfectly into the QbD concept since the process functioning and the product quality are predicted in the first phases of the design”.







Leave A Comment